A mechanistic study of the Knoevenagel condensation reaction: new insights into the influence of acid and base properties of mixed metal oxide catalys ... - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/C7CP04743F

Acid-catalyzed Knoevenagel condensation reaction (synthesis of compound... | Download Scientific Diagram

Catalytic C–H Bond Activation and Knoevenagel Condensation Using Pyridine-2,3-Dicarboxylate-Based Metal–Organic Frameworks | ACS Omega

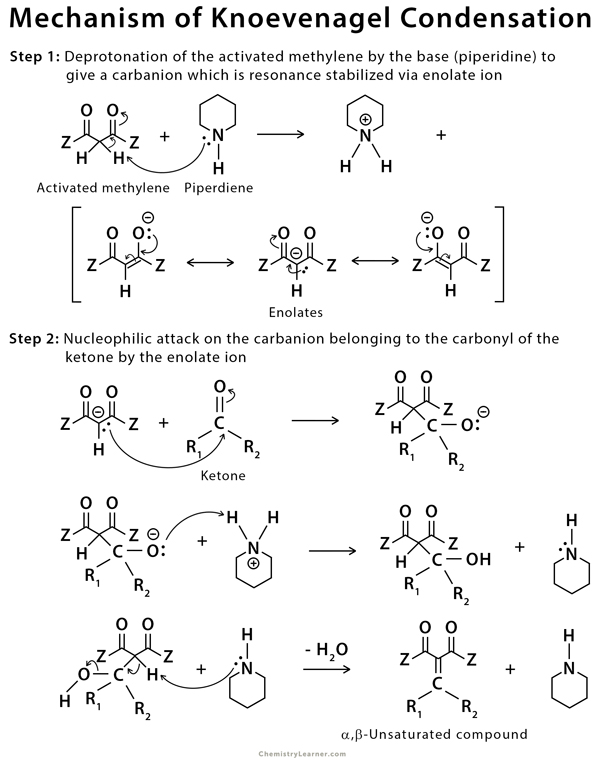
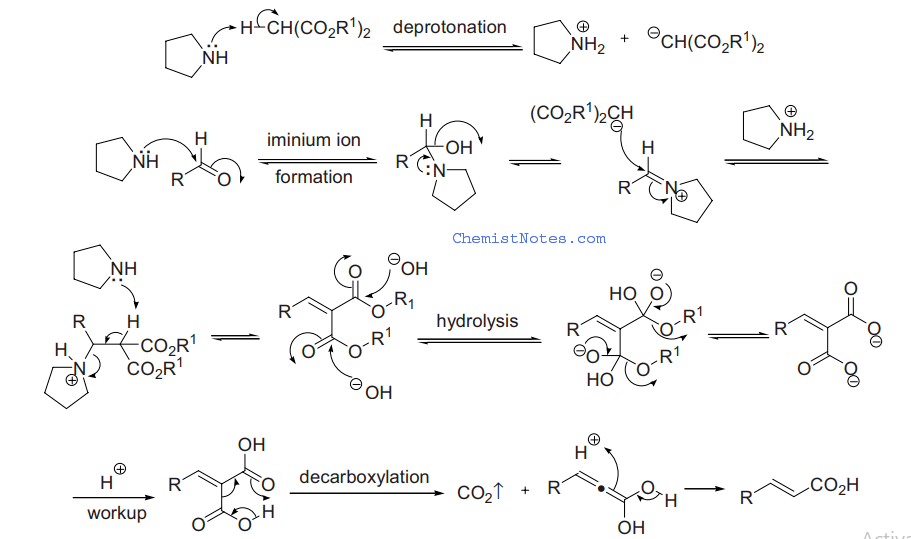
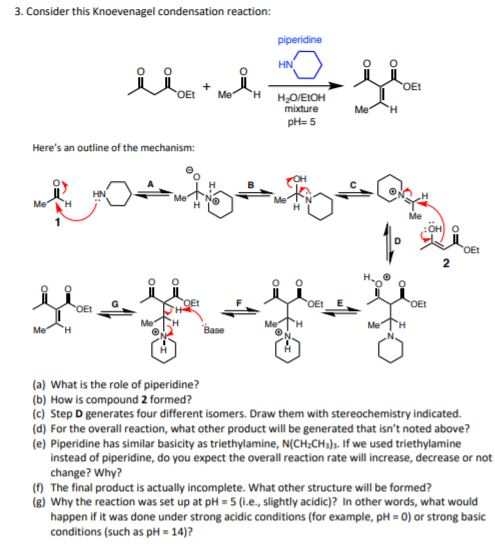
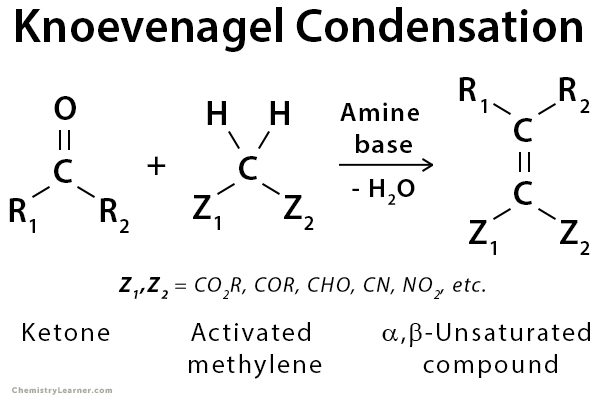
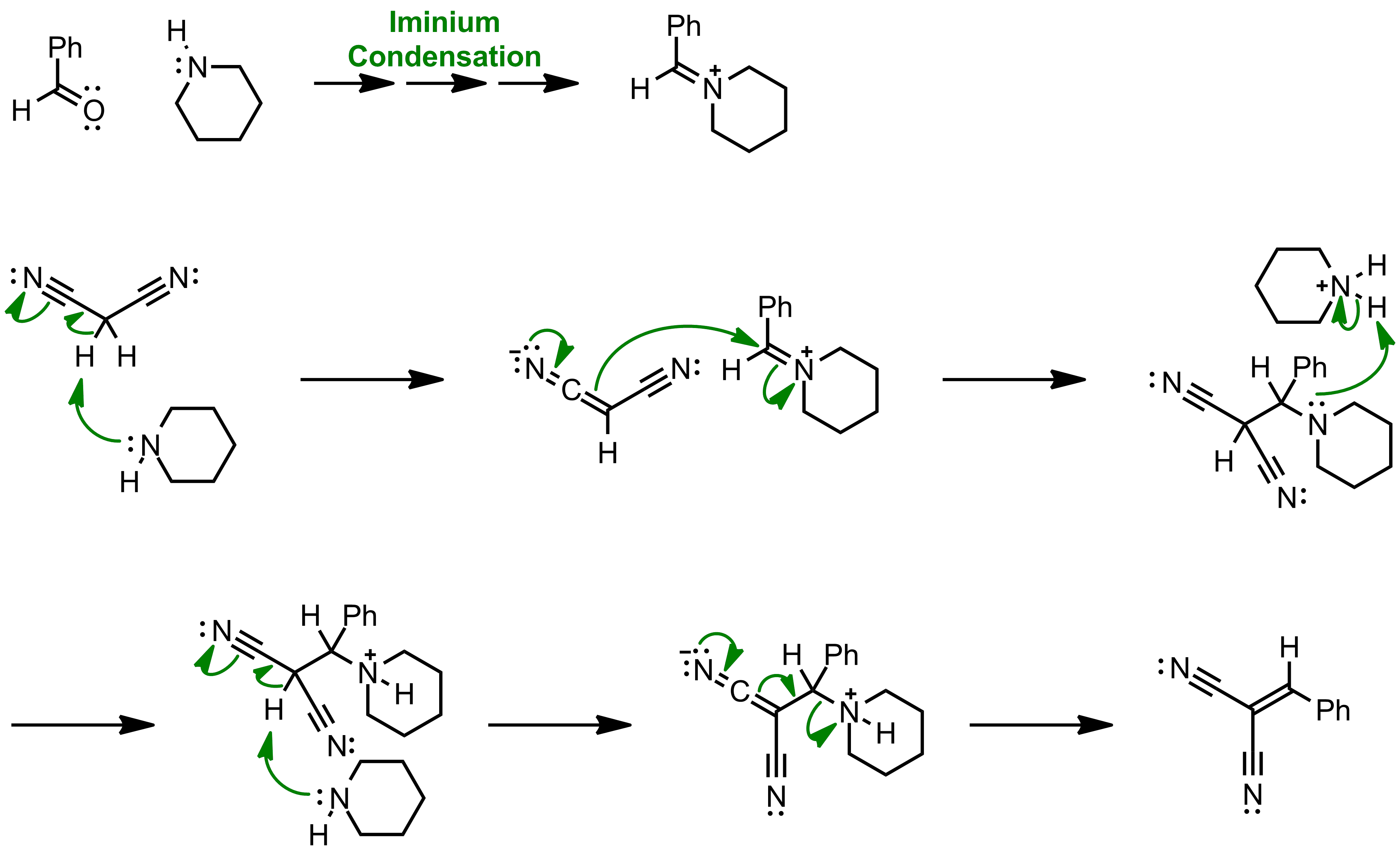
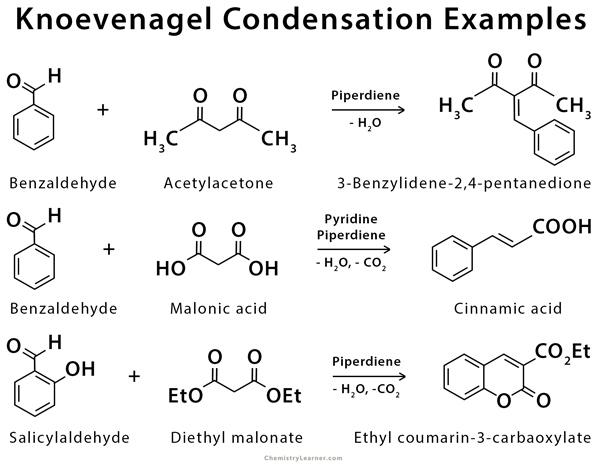
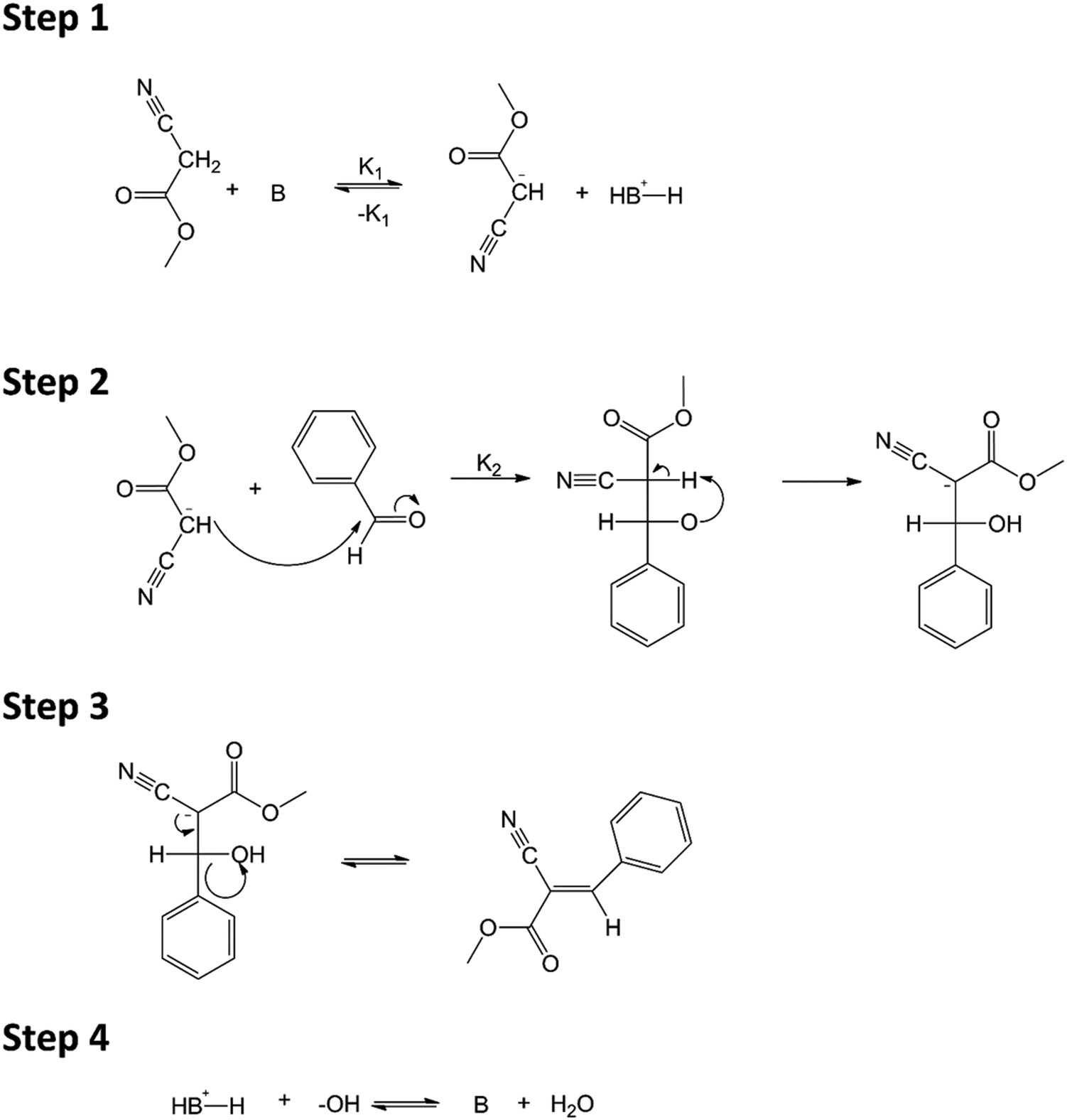
The Knoevenagel reaction is a carbonyl condensation reaction of an ester with an aldehyde or ketone to yield an \alpha , \beta -unsaturated product. Show the mechanism for the Knoevenagel reaction

The Knoevenagel reaction is a carbonyl condensation reaction of an ester with an aldehyde or ketone to yield an \alpha , \beta -unsaturated product. Show the mechanism for the Knoevenagel reaction

Contribution of Knoevenagel Condensation Products toward the Development of Anticancer Agents: An Updated Review - Tokala - 2022 - ChemMedChem - Wiley Online Library